Myths about Aluminium Brazing Fluxes – Part 1
Myth – Fluxes With a Lower Melting Range are Superior
There are claims that a lower melting point flux is better for brazing (i.e. melting between 550 and 560°C – approximately 10 – 15°C below conventional fluxes). The idea here is to try to fool the engineer by illustrating the merits of “early” flux melting, and thus “prolonged” flux action. However, the facts are very different.
As soon as the flux begins to melt, one of the components of the flux – KAlF4 – begins progressively evaporating, with a vapour pressure determined to be 0.08 mbar at 600°C. Evaporation of KAlF4 causes the flux melt to change composition, and it begins to dry out. Given enough time, it is possible for the flux melt to completely dry out before reaching the maximum peak brazing temperature.
A good brazing flux only needs to be available just before filler metal melting. The following table describes what happens at brazing temperature:
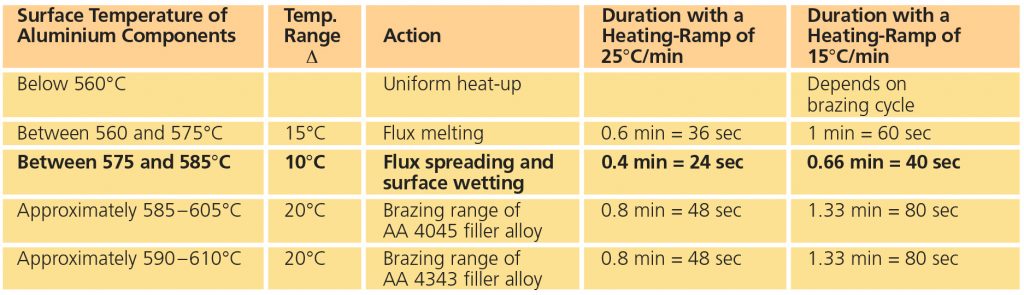
Table 1
As soon as the flux melts, it begins to dissolve the oxide layer, and this solvating process continues until the oxide is removed, even if the filler alloy has melted. The above table shows that even if the period of flux activity would be limited only to the time between complete flux melting and the lower brazing range of AA 4045, it is still adequate. The authors thus consider a flux melting range between 560 and 575°C as the most suitable for aluminium brazing with Al-Si filler alloys.
One should not completely dismiss the point made about “prolonged” fluxing action with lower melting point fluxes. However, once again, all the information must be examined. It has been shown that with an increase in the K2AlF5 content, the flux will start to melt at a lower temperature so that the flux will work at a lower temperature. However, even if KAlF4 evaporation is ignored increasing the K2AlF5 content eventually prevents the flux from spreading smoothly, and therefore affects the efficiency of the flux.
Merely lowering the melting point does not in itself create a better brazing flux.
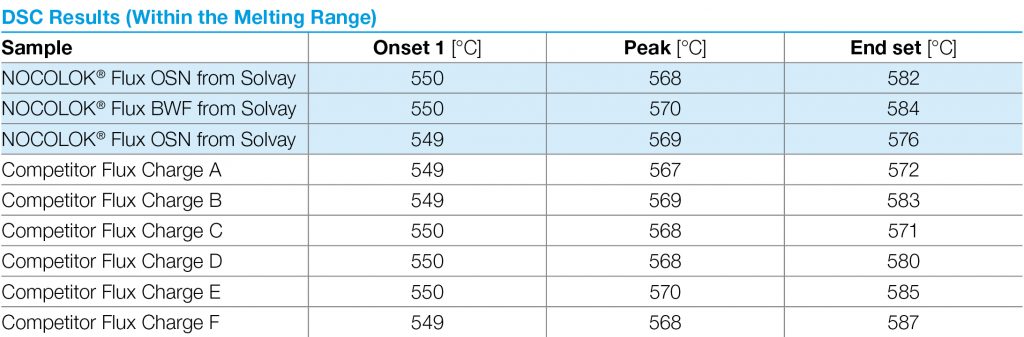
Table 2
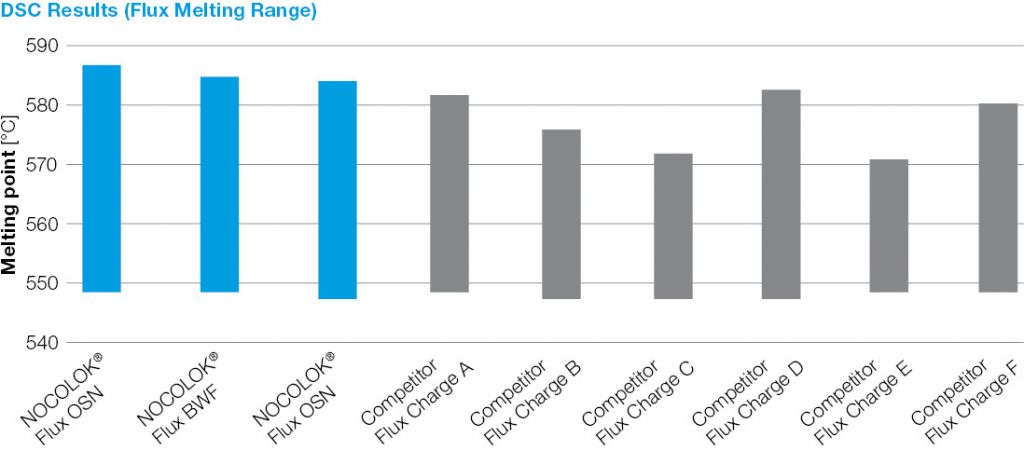
Figure 1

Hinterlasse einen Kommentar
An der Diskussion beteiligen?Hinterlasse uns deinen Kommentar!