Cladding Alloys
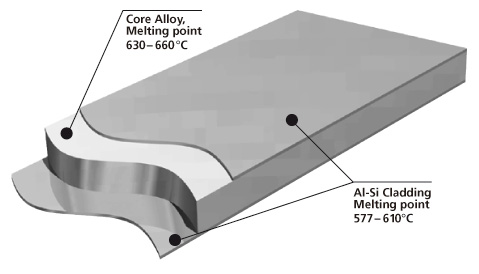
Brazing sheet comprises of a core alloy clad on 1 or 2 sides with a lower melting aluminum-silicon (Al-Si) alloy. This thin layer, usually makes up 5 % to 10 % of the total thickness of the brazing sheet.
It melts and flows during the brazing process, to provide upon cooling a metallic bond between the components. It is common that the braze clad alloy are from the AA 4xxx series or more particularly AA 4343 (Al-6.8~8.2 wt.% Si).
However, if larger fillets are desirable, or if in a situation where brazing is likely to occur at lower temperatures, AA‑4045 is the preferred choice.
Manufacturing

Cross-section morphology of a double-side clad tubestock.
Aluminium brazing sheet is manufactured by roll-bonding techniques to clad a core alloy ingot on one side or both sides with a low melting AlSi alloy. As an alternative, one side can be clad with a non-braze alloy, e.g. Zn-containing alloy.
Depending on the desired final properties, the core is either homogenized or not before the cladding operation.
The whole package is subjected to preheating, hot rolling and cold rolling down to the final thickness of the respective products. Depending on the requested final properties, the material is subjected to final annealing and / or intermediate annealing operation(s).
The core provides structural integrity. It is common to use a variety of aluminium alloys, examples being AA 3xxx (AlMn), more particularly AA 3003, AA 3005, AA 3105 and modified versions of AA 3003 or AA3005 (long life alloys).
Phase diagram
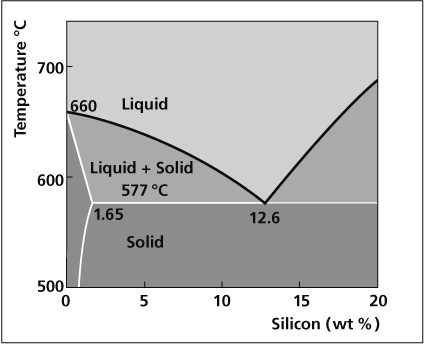
Aluminum End of Al-Si Phase Diagram
The melting characteristics of the cladding alloys are governed by the Al-Si phase diagram. The eutectic composition, i.e. the amount of Si required to produce the lowest melting point is 12.6%. The melting point at this composition is 577°C. At lower Si levels the solidus or the point at which melting begins is also 577°C. However, melting occurs in a range and the temperature above which the filler is completely molten is called the liquidus. In between the solidus and liquidus, the filler is partially molten, existing both as liquid and solid. The difference between the solidus and the liquidus forms the basis for various filler metal alloys.
The table below shows the solidus and liquidus of common brazing alloys.
[table id=2 /]
The higher Si alloys (e.g. AA4047) have higher fluidity and a narrower melting point range while the lower Si alloys have less fluidity with a wider, higher melting point range. Erosion of the base metal occurs when the braze alloy dissolves part of the core alloy. The extent of erosion is increased by:
- Higher Si levels in the braze alloy
- Longer braze cycles
- Excessive peak brazing temperatures
- Excessive thickness fo the braze metal layer
- A design which allows pooling of the braze metal to occur

where can I find the temperature curve for various brazing alloys
Most of alloys do not have a single temperature melting point as it is in case of pure metals, but their melting temperature is between a certain temperature interval called melting range. It means that when an alloy is at temperature within its melting range only part of its volume is liquid and part is solid and the system as long as the temperature does not change, remains at this equilibrium.
The information about melting range of a given alloy is fundamental for a designer of a part which is to be brazed. Simply speaking the melting range of the joined parts must be higher than that of the filler alloy.
An excellent source to look for the melting range of an alloy is a website called MatWeb http://www.matweb.com/ . One should however remember that this site works well when international designations are used. Designations used by individual suppliers will not yield positive search results. In such case information about the melting range should be obtained from the aluminum supplier.
Hi, Neat post. There is a problem with your website in internet explorer, would check this… IE still is the market leader and a good portion of people will miss your magnificent writing because of this problem.
Dear Reader, we checked the compatibility of this site.
It works fine with IE 6, IE 7 and IE 8.
Earlier versions of IE are not supported.
Hello Solvay
As described, the five factors will increase the extend of erosion, can you share the reaction mechanism?
Thank
Jason
Dear Jason,
I would like to answer your question – with a quote from
http://www.alueurope.eu/wp-content/uploads/2012/01/AAM-Joining-4-Brazing.pdf (point 4.2.5):
Dissolution of core alloy
Dissolution of aluminum core alloy is the local melting of the core alloy in contact with the filler metal. It is caused by diffusion of Silicon from the filler metal into the core alloy (grain boundaries first). The melting point of this area is thus lowered, and it melts down. This phenomenon must be minimized, because it affects the strength and corrosion resistance of the product. It can even lead to a perforation of the core alloy.
Aluminum core alloy dissolution is affected by time and temperature – and by the Si level and quantity in the liquid filler alloy.
I hope this information is helpful. Don’t hesitate to contact me again, if i can be of further assistance to you.
Can you please let me know for which company you work – and at which location? There might be a local technical expert from Solvay, which I could connect you with.
Thank you.
Best regards,
Hans Swidersky
Nice info Dr. would like to know whether the core alloy dissolution is recommended? any international standard available for its acceptance.
Dear Hans,
your article is short & crisp. I recently getting into these product lines in our company…which is one among top suppliers for thermal products.
My question :
1) Why erosion takes place when silicon dissolves in the core metal ?
2) is higher melting range is desirable or vice versa for good brazing ?
3) Is 12.6 % silicon is ideal for good brazing ?
Also if you have any other training resource to understand about brazing , please forward to my email ID.
Thank you
Gowrisankar
Dear Gowrisankar,
1. Silicon diffusion into the core metal results in formation of Al-Si alloy, which has lower melting rage than the original core metal. Thus, we are facing two effect: – change of alloy compositiwith on and structure, and – change (i.e. reduction) of melting range.
2. I am not sure if I understand this question correctly. What is „higher melting range“ referring to: – the filler alloy or – the core material?
3. 12.6% Si in Al is the so-called ‚eutectic‘ composition - i.e. the Al-Si alloy composition with the lowest melting range of this combination. The specified melting range of AlSi 12.6 (AA 4047) is between 577 and 583°C.
This alloy is used in flame brazing and in induction brazing.
For furnace brazing (i.e. controlled atmosphere brazing with non-corrosive flux), the filler alloys of clad aluminium sheet is either AlSi7 (AA 4343) or AlSi10 (AA 4045).
Filler alloy selection will depend on the specific application and the design of the parts to be joined.
Training Resource:
Please refer to out technical brochures and publications – via: https://www.aluminium-brazing.com/sponsor/nocolok/Html/Downloads/index.html.
Best regards,
Hans Swidersky
HI,
looking to optimize a design for dip brazing. Where can i find a supplied of clad material with a core of some easy to machine AL alloy like 6061-T6?
Thanks
Bill
Hello
we are looking for Alsi12 material in sheet strip or coi with thickness 1.1,5; 2; 2.5; 3 mm
Do you know a supplier or were we could find?
Best regards
Alfredo
What specification defines Number 14 clad sheet Grades 4004-6951-4004?
I find it interesting when you said that the core of the metal clad has to be either homogenized or not depending on the final properties you wanted for them. I can imagine how certain materials would have various processes to undergo before they are put out into the market and before they are installed. It just shows how important it is to have professionals do the installations for such materials to ensure that they are done right, especially when every project can have different requirements.