Brazing of Aluminium Alloys withHigher Magnesium Content using Non-Corrosive Fluxes – Part 3
Technical Information by Leszek Orman, Hans-Walter Swidersky and Daniel Lauzon
Abstract
For just as long as aluminium has been used for brazing heat exchangers, there has been a trend to down-gauging components for weight savings. The most common alloying element to achieve higher strength alloys for the purpose of down-gauging is magnesium. While magnesium additions are helpful in achieving stronger alloys, the consequence is a decrease in brazeability. This article discusses the mechanism of brazing deterioration with the addition of magnesium and proposes the use of caesium compounds as a way of combating these effects.
We split the article in five parts:
- Introduction
- Effects of Mg on the Brazing Process
- Mechanism of Magnesium Interaction with the Brazing Process
- Caesium Fluoroaluminates
- NOCOLOK® Cs Flux
Mechanism of Magnesium Interaction with the Brazing Process
According to M. Yamaguchi et al [5], when magnesium diffuses to the surface during brazing, a chemical reaction takes place with the flux resulting in the generation of KMgF3.
The authors suggest the following equations to explain some of the chemical interactions between magnesium and K1-3AlF4-6 flux:
- 3 MgO + 2 KAlF4 → MgF2 + 2 KMgF3 + Al2O3 (a)
- 3 MgO + 2 KAlF4 → 2 MgF2 + K2MgF4 + Al2O3 (b)
- 3 MgO + 2 K3AlF6 > → 3 K2MgF4 + Al2O3 (3)
By performing XRD (X-ray Diffraction) phase identification on products brazed with Mg containing alloys, A. Gray et al [6] confirmed the presence of K2MgF4, spinel oxide (Al2MgO4) and possibly KMgF3. These magnesium containing compounds have a characteristic needle like morphology as shown in Fig. 5.
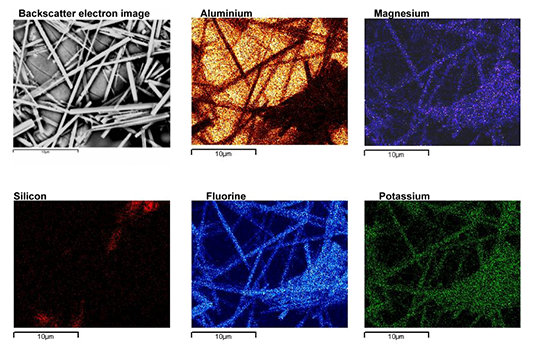
Fig. 5: Morphology of magnesium containing compounds as seen by Scanning Electron Microscope [6].
H. Johansson et al [7] also determined that at temperatures above 425°C the magnesium diffusion to the surface is very rapid resulting in the formation of magnesium oxide (MgO) and spinel oxides (Al2MgO4). These oxides have very low solubility in NOCOLOK® Flux. Subsequently these magnesium oxides react with the flux resulting in the formation of magnesium fluoride (MgF2) and potassium magnesium fluorides (KMgF3, K2MgF4, see equations a), b), and c)). These reactions change the flux chemical composition causing its melting range to rise. The melting point of these magnesium fluorides is very high, which in turn drives the melting point of the flux upwards, thereby decreasing the activity of the flux. The above factors also cause a decrease in the flowing characteristics of the flux thus lowering its overall effectiveness. Therefore the desired key point to limit the flux poisoning effect would be to reduce the formation of magnesium oxides and potassium magnesium fluorides.
Download the complete article as a PDF-File.
References:
- S. W. Haller, “A new Generation of Heat Exchanger Materials and Products”, 6th International Congress “Aluminum Brazing” Düsseldorf, Germany 2010
- R. Woods, “CAB Brazing Metallurgy”, 12th Annual International Invitational Aluminum Brazing Seminar, AFC Holcroft, NOVI, Michigan U.S.A. 2007
- T. Stenqvist, K. Lewin, R. Woods “A New Heat-treatable Fin Alloy for Use with Cs-bearing CAB flux” 7th Annual International Invitational Aluminum Brazing Seminar, AFC Holcroft, NOVI, Michigan U.S.A. 2002
- R. K. Bolingbroke, A. Gray, D. Lauzon, “Optimisation of Nocolok Brazing Conditions for Higher Strength Brazing Sheet”, SAE Technical Paper 971861, 1997
- M. Yamaguchi, H. Kawase and H. Koyama, ‘‘Brazeability of Al-Mg Alloys in Non Corrosive Flux Brazing’’, Furukawa review, No. 12, p. 139 – 144 (1993).
- A. Gray, A. Afseth, 2nd International Congress Aluminium Brazing, Düsseldorf, 2002
- H. Johansson, T. Stenqvist, H. Swidersky “Controlled Atmosphere Brazing of Heat Treatable Alloys with Cs Flux” VTMS6, Conference Proceedings, 2002
- U. Seseke-Koyro ‘‘New Developments in Non-corrosive Fluxes for Innovative Brazing’’, First International Congress Aluminium Brazing, Düsseldorf, Germany, 2000
- K. Suzuki, F. Miura, F. Shimizu; United States Patent; Patent Number: 4,689,092; Date of Patent: Aug. 25, 1987
- L. Orman, “Basic Metallurgy for Aluminum Brazing”, Materials for EABS & Solvay Fluor GmbH 11th Technical Training Seminar – The Theory and Practice of the Furnace and Flame Brazing of Aluminium, Hannover, 2012
- K. Suzuki, F. Miura, F. Shimizu; United States Patent; Patent Number: 4,670,067; Date of Patent: Jun. 2, 1987
- J. Garcia, C. Massoulier, and P. Faille, „Brazeability of Aluminum Alloys Containing Magnesium by CAB Process Using Cesium Flux,“ SAE Technical Paper 2001-01-1763, 2001

Hinterlasse einen Kommentar
An der Diskussion beteiligen?Hinterlasse uns deinen Kommentar!