The Theory and Practice of the Flame- and Furnace-Brazing of Aluminium
Dates: September 6 & 7, 2016 in Hannover/Germany

Purpose of the Seminar:
This technical training seminar will be presented in the English language at the Conference Centre and laboratories of Solvay GmbH, in Hannover, Germany. It will provide information concerning the manufacturing practices commonly used for brazing operations and, in particular, will address the three fundamental aspects of the industrial-scale brazing of aluminium. These are:
- The flame brazing of aluminium.
- Controlled Atmosphere Brazing (CAB) of aluminium heat exchangers with non-corrosive fluxes (NOCOLOK® Flux).
- The methodology of how to ensure that the brazing process selected is, indeed, the one that represents ‘best practice’.
Who should attend this two-day seminar?
- Technical staff who need to have a specific understanding of either one or both of the fine details of the technology of the brazing of aluminium with flames, and/or the NOCOLOK® furnace brazing process.
- Design and production engineers who are fabricating, or who are intending to fabricate, aluminium pipe-work assemblies and/or condensers and/or evaporators.
- Production Engineering Department Managers whose duties include day-to-day responsibility for the brazing of aluminium.
Watch the video of the EABS Technical Training Seminar
Here you can find the detailed seminar programme and registration.




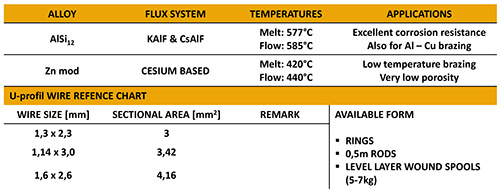
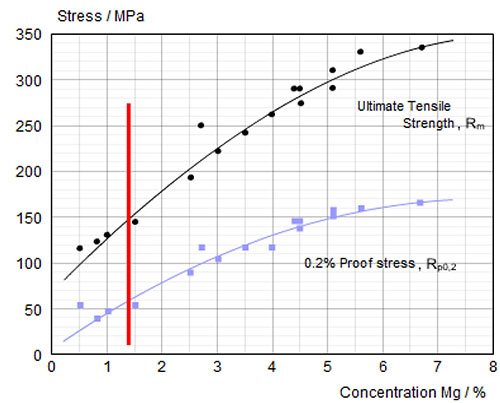

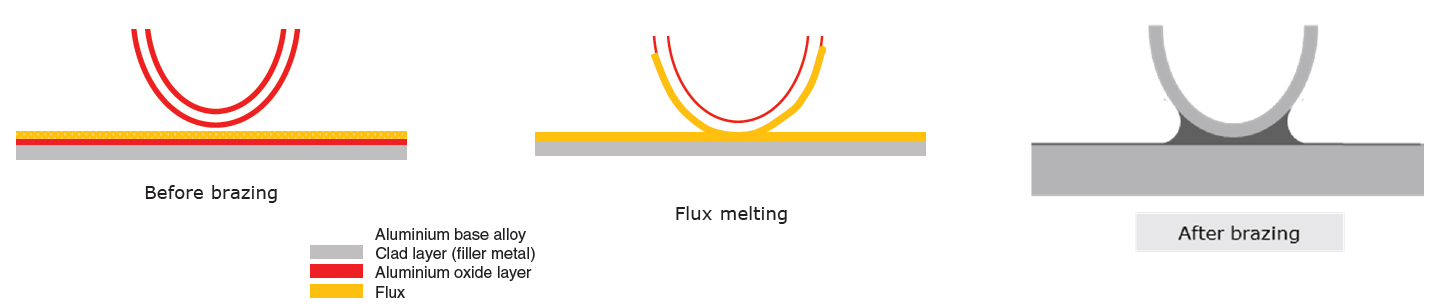





 flame brazing of aluminium to copper.
flame brazing of aluminium to copper.